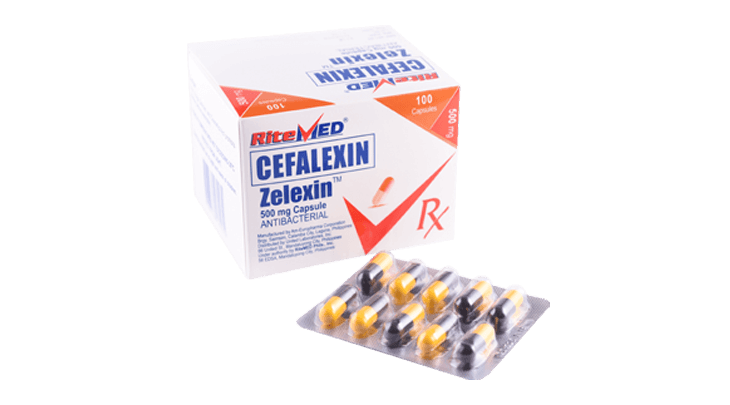
Product Details
What is this medicine used for?
For the treatment of the following infections cause by susceptible microorganisms: Upper respiratory tract infection including otitis media, pharyngitis, tonsilitis, and sinusitis. Lower respiratory tract infection including pnuemonia, acute bacterial exacerbations of chronic bronchitis, and secondary bacterial infections of acute bronchitis. Uncomplicated skin and skin structure infections. Urinary tract infections, inlcuding pyelonephritis and cystitis.
How much and how often should you use this medicine?
Usual Pediatric Dose: Orally, 20 mg/kg body weight/day in divided doses every 8 to 12 hours, depending on the type and severity of infection. Bronchitis and pneumonia: 20 mg/kg body weight/day in divided doses every 8 hours. Pharyngitis, tonsilitis, skin and skin structure infections: 20 mg/kg body weight/day in divided doses every 12 hours. In more serious infections, otitis media and infections caused by less susceptible organisms, 40 mg/kg body weight/day in divided doses every 8 to 12 hours. Maximum dose: 1 g/day.
Contraindication:
Known hypersensitivity to cephalosporins, or any component of the product.
Warnings and Precautions:
Careful inquiry should be made prior to cefaclor therapy to determine whether the patient has had
previous hypersensitivity reactions to cefaclor, cephalosporins, penicillins, or other drugs. Use with
caution in penicillin-sensitive patients since cross-hypersensitivity among β-lactam antibiotics has
been clearly documented and may occur in up to 10% of patients with a history of allergy to penicillin.
In case of an allergic reaction to cefaclor, the drug should be discontinued. Serious acute hypersensitivity reactions require immediate emergency treatment with antihistamines, corticosteroids, pressor amines, and airway management, including intubation, should
also be instituted.epinephrine. Oxygen, intravenous (IV), IV antihistamines, corticosteroids, pressor amines, and airway management, including intubation, should
also be instituted. Antibiotics, including cefaclor, should be administered cautiously to any patient who has demonstrated some form
of allergy, particularly to drugs. Clostridium difficile associated diarrhea (CDAD) has been reported with use of nearly all antibacterial
agents, including cefaclor, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile. C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producingstrains of C. difficile caused increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibiotic use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents. If CAD is suspected or confirmed, ongoing antibiotic use not directed againts C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein suplementation, antibiotic treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated. Use with caution in patients with markedly impaired renal function.
Antibiotics, including cephalosporins, should be prescribed with caution in individuals with a history of
gastrointestinal disease, particularly colitis.
Prescribing cefaclor in the absence of a proven or strongly suspected bacterial infection or a prophylactic
indication is unl ikely to provide benefit to the patient and increases the risk of the development of drug-resistant
bacteria.
As with other antibacterial drugs, long term or repeated use may result in overgrowth of non-susceptible
organisms, including fungi.
Effects on ability to drive and use machines
Cefaclor is presumed to be safe or unlikely to produce an effect.
Undesirable Effects:
Infections and other infestations: Psuedomembranous colitis. Blood and lymphatic system disorders: Eosinophilia, thrombocytopenia, leukopenia, lymphocytosis, neutropenia, prothrombin time increased, hemolytic anemia, agranulocytosis, aplastic anemia. Immune system disorders: angioedema, anaphylaxis, (maybe manifested by solarity symtoms, including asthenia, edema of the face and limbs, dyspnea, paresthesias, hypotension, vasolidation), urticaria. Nervous system: Headache, dizziness, somnolence, seizures. Psychiatric disorder: somnolence, reversible hyperactivity, nervousness, insomia, confusion, hallucinations. Gastrointestinal disorders: Diarrhea, nausea, vomiting, dyspepsia. Hepatobiliary disorders: transient elevation in aspartate in aminotrasnferase (AST), alanine aminotransferase (ALT) and alkaline phosphetase, hepatic dysfunction, including hepatitis and cholastic jaundice. Skin and subcautineous tissues disorders: rash, pruritus, serum sickness like reaction (charatcterized by erythema multiforme, rashes, and other skin manifestations accompanied by arthritis/arthralgia, with or without fever, and differ from classic serum sickness in that there is frequently associated lymphadenopathy and proteinuria, or circulating immune complexes, and no evidence to date of sequelea of the reaction), Steven-Johnson syndrome, toxic epidermal necrolysis, morbiliform eruptions. Musculoskeletal and connecitive tissue disorders: Hypertonia. Renal and unrinary disorders: Transient increase in serum urea or creatinine, abnormal urinalysis, reversible interstitial nephritis. Reproductive system and breast disorders: Vaginal monilliasis, vaginitis. General disorders and administrations site conditions: Fever. In addition to the adverse reaction listed above, the following adverse reactions and altered laboratory tests have been reported for cephalosporin-class antibiotics: fever, abdominal pain, superinfection, renal dysfunction, toxic nephropathy, hemorrhage,false positive test for urinary glucose, elevated bilirubin, elevated lactate dehydrogenase (LDH), and pancytopenia.
Interaction w/ other medicaments:
Antacids: the extent absorption of cefaclor is diminished if magnesium or aluminum hydroxide- containing anatacids are taken an hour of administration; cimetidine did not alter either the rate of the extent of the absorption of cefaclor. Anticoagulants: concomitant administration of cefaclor and oral anticoagulants has rarely resulted in increased prothrombin time, with or without clinical bleeding. It is recommended that during concomitant treatment, regular monitoring of prothrombin time should be considered, with dosage adjustment if necesarry. Probenecid: As with other beta-lactam antibiotics, the renal excretion of cefaclor is inhibited by probenecid. Interference with laboratory test: Cefaclor may cause false positive reaction for glucose in the urine with Benidict's solution. Fehling's solution or with Clinitest tablets, but not with enzyme-based tests such as Clinistix. A false-positive Coomb's test has been reported during treatment with the cephalosporins antibiotics; therefore, it should be recognized that a positive Coomb's test may be due to the drug, e.g., Coomb's testing of newborns whose mothers have received cephalosporins before parturition or in hematologic studies, or in transfusion cross-matching procedures when antiglobulin tests are performed.